The Imathera Platform
IMATHERA’s research projects are structured around four major areas: cancerology, pulmonary fibrosis, cardiovascular disease and preclinical imaging methodology.
Together, let’s overcome cancer.
Imathera Platform
Molecular imaging has become a crucial tool for the detection and treatment of cancers for many years. Since 2011, the Nuclear Medicine Department of the Georges-François Leclerc Center has been involved in translational imaging research by hosting the IMATHERA platform, which provides a comprehensive approach to pharmacoimaging and radiation therapy in an exclusively dedicated setting.
Thanks to its strategic location within the Dijon university campus and its membership in the Pharm’image® GIE, the CGFL works with numerous highly specialized local partners in complementary fields ranging from the design of molecules for radioimaging (ICMUB, CheMatech) to the processing of images taken on patients (CGFL, Dijon University Hospital) and preclinical validation stages (Oncodesign, IMATHERA), which constitutes a unique local field of expertise. Its proximity to eminent research centers (INSERM, UB, CNRS) and clinicians allows IMATHERA to be part of a translational research approach dedicated to patients.

Our main partners


Our means
The preclinical imaging platform, within the Nuclear Medicine Department headed by Professor Alexandre Cochet (PU-PH), features a 575 m² laboratory entirely dedicated to preclinical imaging and is accredited by the French Nuclear Safety Authority (ASN) and the Departmental Directorate for Population Protection (DDPP). Its highly specialized staff includes a multidisciplinary team led by Dr. Bertrand Collin , a radiopharmacist and lecturer-researcher responsible for the platform; Dr. Alexandra Oudot , deputy head; Dr. Pierre-Simon Bellaye , head of research; Dr. Alexandre Dias and Dr. Romain Douhard , researchers; and Alex Helbling , study director. The team also includes a radiophysicist (Dr. Jean-Marc Vrigneaud ), two radiation protection specialists (Lydie Houot and Mickaël Pichon), and a PET/MRI imaging researcher (Dr. Alan Courteau ). IMATHERA’s work relies on a highly specialized technical team ( John Simonet and Mélanie Guillemin ) and students (Romain Fontaine-Tuffery, Agnieszka Kownacka, Sarah Jedidi, Jame Frenay and Camille Petitot , doctoral students).
Thanks to the radiochemistry, radiolabeling and quality control laboratories, it is possible to design innovative imaging probes based on the latest generation of equipment. The molecular probes are then evaluated in vitro and in vivo using a complete range of equipment that allows the detection technology to be adapted to the biological problem: scintigraphy camera coupled with a scanner, PET (positron emission tomography) coupled with a scanner, PET coupled with MRI (magnetic resonance imaging), optical imaging, or 2D imaging.
Since 2012, IMATHERA has hosted the Equipex “Investments for the Future” program IMAPPI (Integrated Magnetic Resonance and Positron Emission Tomography in Preclinical Imaging), funded by the French National Agency for Research (ANR) to the tune of €7.3 million and led by Professor François Brunotte (PU-PH). This project aims to develop a new preclinical imager, unique in Europe, combining the high sensitivity of PET with the high resolution of MRI. This technology is currently available in very few locations worldwide and only in prototype form. PIRP also receives funding from public institutions (ANR, Bourgogne Franche-Comté region, etc.) and associations (League Against Cancer, Fondation d’avenir, ARC, Fonds de Recherche en Santé Respiratoire, etc.) to carry out its research projects.
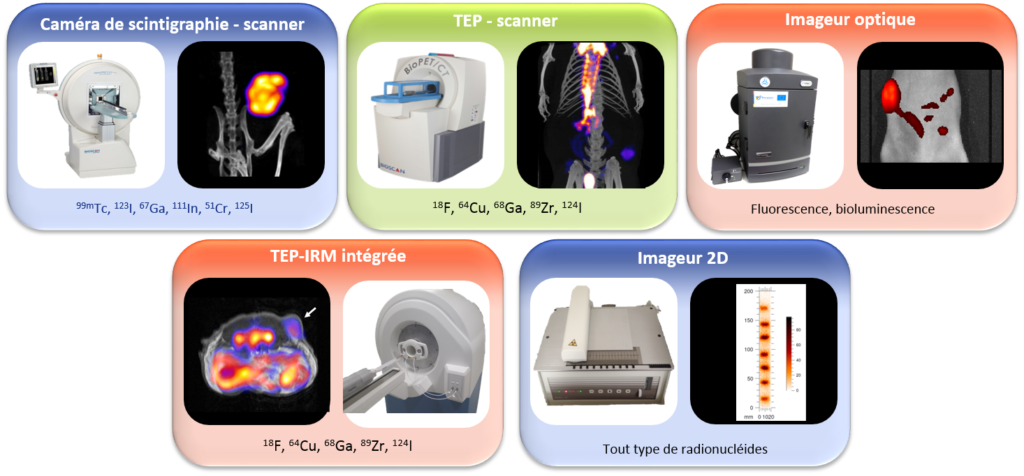
In each study conducted on the platform, particular attention is paid to getting as close as possible to the clinical conditions encountered in patients by adapting the developed imaging probes as closely as possible to living organisms. To achieve this, models that are as close as possible to human pathology are used using the same imaging modalities as in the nuclear medicine clinical department.
Our main projects
CANCEROLOGY
VECTORIZED INTERNAL RADIOTHERAPY – The COMETE project
The COMETE project (moleCular radiotherapy for METastatic Colorectal and gastric cancErs) responds to the FEDER-FSE+ regional call for projects. It focuses on the development of a portfolio of vectorized internal radiotherapy (RIV) molecules for the diagnosis and treatment of advanced digestive cancers. The project aims to develop two RIV candidates up to preclinical dosimetry studies, just before moving on to clinical evaluation. The COMETE project is led by a consortium consisting of the company Oncodesign Precision Medicine (OPM), the Institute of Molecular Chemistry of the University of Burgundy (ICMUB, UMR CNRS 6302) and the CGFL, all based in Dijon.

Internally Directed Radiotherapy (IDR) is a breakthrough therapeutic innovation for the treatment of certain metastatic cancers. It involves administering small molecules carrying a radioactive atom to patients, which will recognize and bind to a “target” expressed or overexpressed by tumor cells. The radioactivity delivered directly to the tumor cells will cause damage to their DNA and thus stop the progression of the disease. These treatments are administered under the responsibility of nuclear medicine physicians, in close collaboration with medical oncologists.
THE VIR IN 4 ACTS :
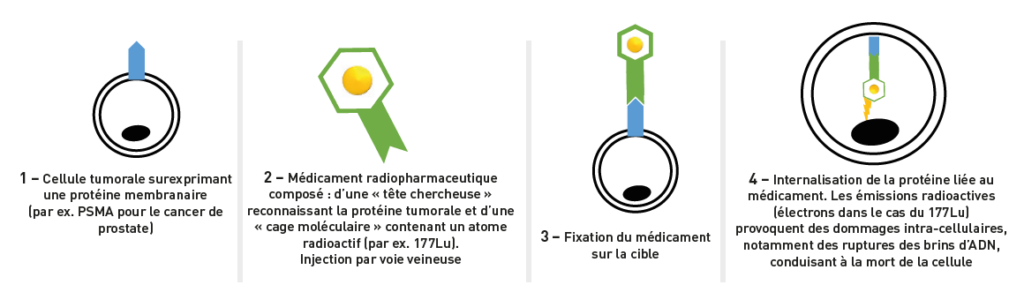
The other major advantage of IVR is the possibility of combining these treatments with “companion” diagnostic tools: the same molecule that targets the tumor can be used to obtain Positron Emission Tomography (PET) images, making it possible to verify that the target is indeed expressed by the tumors. This is called a “theranostic” approach (“thera” being the contraction of therapy and “nostic” the contraction of diagnostic). These images taken before IVR will make it possible to predict the effectiveness of the treatment, and therefore to only offer it to patients likely to benefit from it. This theranostic strategy, which also allows the monitoring of the effectiveness of IVR treatments, is therefore part of a personalized medicine approach.
Digestive cancers represent more than 20% of cancers in France, among which colorectal, gastric and pancreatic cancers correspond to 61%, 12% and 8% respectively. Colorectal cancer is the 2nd most diagnosed cancer in women and the 3rd in men. Surgery is the mainstay of treatment for local and loco-regional pathologies. However, nearly 50% of patients with colorectal cancer will develop metastases, which contributes to a high mortality rate in the face of the failure of current therapies. The rise of new therapeutic strategies for colorectal cancer, combining surgery, radiotherapy and chemotherapy, has not led to a significant improvement in the 5-year survival of metastatic patients, which remains below 10%.
Gastric cancer is the 5th most commonly diagnosed cancer and the 3rd leading cause of death worldwide. The prognosis of gastric cancer is linked to the stage of the disease at the time of diagnosis. The therapeutic offer for advanced gastric cancer, characterized by the appearance of metastases, is very limited. In particular, the presence of cerebral metastases worsens the prognosis and the average median survival is between 1.3 and 2.4 months.
Pancreatic cancer is the 6th most commonly diagnosed cancer in France and the 4th leading cause of death in women and the 5th leading cause of death in men. The 5-year survival rate for patients with pancreatic cancer is less than 4%, making it a cancer with a very poor prognosis.
In this project, we propose to develop treatments by vectorized internal radiotherapy (RIV), making it possible to meet this unmet clinical need and to increase the survival of patients with metastatic digestive cancers.
The COMETE project is structured around 4 main scientific objectives:
- The identification and validation of tumor targets in metastatic digestive cancers, via artificial intelligence approaches and innovative technological platforms for the production of highly specific biological vectors.
- The development of new RIV molecules to treat metastatic digestive cancers, from their design and optimization to the selection of the best molecule for early-phase human development.
- Evaluation of the anti-tumor efficacy and toxicity of RIV molecules on preclinical models of metastatic colorectal and gastric cancers.
- The development of “companion” imaging agents for IVR, to enable the personalization of therapeutic management (selection of responding patients, dose optimization, monitoring of efficacy).
Solid Tumor – TRIPLE Negative Breast Cancer
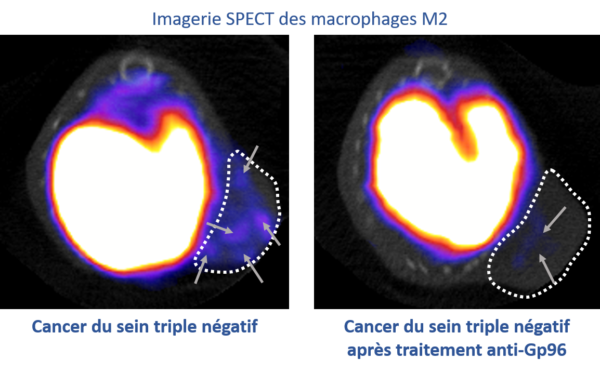
Among the different types of breast cancer, triple-negative breast cancer consists of tumor cells that do not express the receptors targeted by most of the innovative therapies currently available for other types of breast cancer. This cancer is particularly aggressive and associated with high mortality with a 5-year survival rate of less than 25%. Its treatment remains a real challenge today because no targeted therapy can be offered. Triple-negative breast cancer is characterized by an infiltration of immunosuppressive cells which, by suppressing anti-tumor immune responses, promote tumor progression and the development of metastases. Our project therefore aims, on the one hand, to develop new drugs targeting these immunosuppressive cells and, on the other hand, to validate markers for therapeutic monitoring of such treatments.
Our team is currently working in collaboration with the INSERM HSP-pathies team led by Dr. Carmen Garrido on a stress protein: Gp96, whose increased presence in certain tumors is associated with a poor prognosis. We have shown that Gp96 is present on the membrane of a type of immunosuppressive macrophage: M2 macrophages, whose expression is correlated with a poor prognosis. We were able to image these immunosuppressive macrophages in vivo using SPECT imaging in our triple-negative breast cancer models and monitor their reduction when using a Gp96 inhibitor. Thus, imaging these immunosuppressive macrophages could be an innovative tool as a potential marker for prognosis, therapeutic prediction, and/or monitoring in triple-negative breast cancer.
Related publications:
- Bouchard A, Sikner H, Baverel V, Garnier AR, Monterrat M, Moreau M, Limagne E, Garrido C, Kohli E, Collin B, Bellaye PS. The GRP94 Inhibitor PU-WS13 Decreases M2-like Macrophages in Murine TNBC Tumors: A Pharmaco-Imaging Study with 99mTc-Tilmanocept SPECT. Cells. 2021 Dec 2;10(12):3393
- Chaumonnot, S. Masson, H. Sikner, A. Bouchard, V. Baverel, P. S. Bellaye, B. Collin, C. Garrido, E. Kohli, The HSP GRP94 interacts with macrophage intracellular complement C3 and impacts M2 profile during ER stress. Cell Death Dis 12, 114 (2021).
- 99mTc-Timanocept Spect Imaging As A Potential Non-Invasive Method To Quantify CD206+ Tumor-Associated M2-like Macrophages, EMIM 2021 (poster)
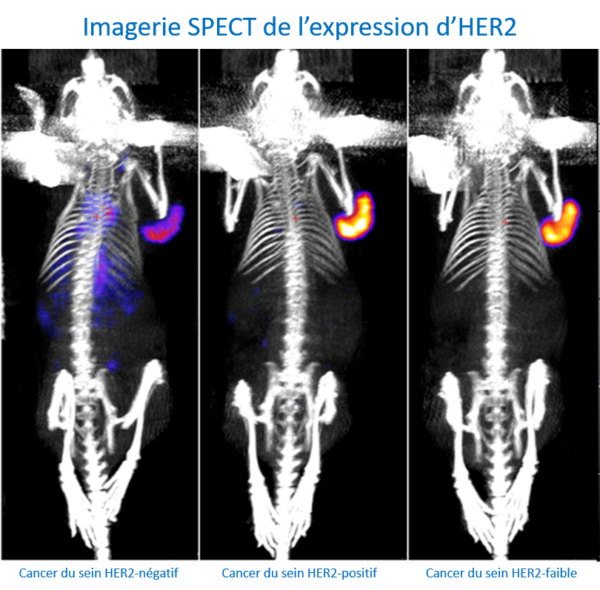
Solid Tumor – Low-HER2 Breast Cancer
Breast cancer is the most common cancer worldwide. In fact, breast tumors in women alone account for approximately 11.7% of new cancer cases each year, with more than two million women diagnosed in 2020, making this disease a major public health issue. At the molecular level, this malignant tumor is highly heterogeneous and can be divided into five groups (Luminal A, Luminal B HER2-negative, Luminal B HER2-positive, HER2-enriched, triple-negative) based on the immunohistochemical expression of estrogen receptors, progesterone receptors, and human epidermal growth factor receptor 2 (HER2). Approximately 20% of newly diagnosed breast cancer cases are HER2-positive, and the prognosis for this group has improved with anti-HER2 targeted therapy based on monoclonal antibodies. Despite this progress, a high percentage of patients (40-50%) have low HER2 expression (HER2-low), in whom anti-HER2 therapies are not very effective. However, recent studies have shown that HER2-low patients may benefit from new HER2-targeted therapies based on “armed” antibodies (antibody drug conjugates, ADCs). However, resistance to these new therapies has been observed. In addition, accurate determination of HER2 expression is essential to identify patients who may benefit from
Our preliminary results showed that nuclear imaging, using a HER2-specific probe, could detect the level of HER2 expression in various preclinical breast cancer models. These results enabled our platform to obtain ANR funding (EITHER2BC) of €324,000 over 3 years (2023-2026) to develop these probes for the diagnosis and therapy of HER2-low breast cancers.
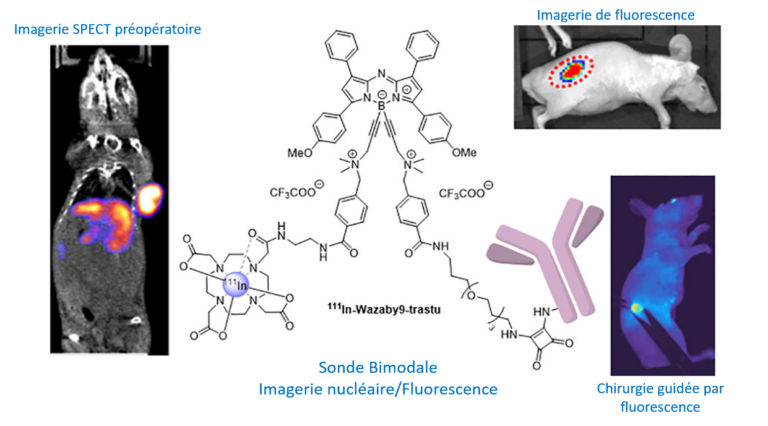
Solid Tumor – Fluorescence-Guided Surgery
Despite the emergence of targeted therapies, surgery still plays a vital role in the treatment of colorectal and pancreatic cancers. A major challenge is the difficulty of detecting and visualizing tumor margins. Fluorescence-guided surgery (FGS) allows surgeons to better visualize anatomical structures in real time. Additionally, FGS can be enhanced by combining preoperative imaging to accurately localize tumors. In this project, we propose to design and evaluate bimodal imaging conjugates (nuclear/fluorescence imaging) in order to improve pre-operative staging (SPECT), intra-operative detection of tumors and their resection (FGS) but also to improve post-operative monitoring (SPECT). Our probes will be tested for FGS in different preclinical models mimicking human pathology, such as orthotopic models of colon and pancreatic cancer, but also models mimicking the formation of liver metastases or peritoneal carcinomatosis. The expected results of this project are, a fine visualization of tumors before surgery (SPECT imaging) and a more precise detection by fluorescence to minimize the extent of resection. Finally, the absence of recurrence can be detected using this compound a few weeks after surgery (SPECT).
This project allowed our platform to contribute to a project funded by INCA (PLBIO23-023) to the tune of 593,460 euros over 3 years (2023-2026) led by Dr. Catherine Paul (École Pratique des Hautes Études – Université PSL) and in collaboration with ICMUB (Dr. Goze Christine) and the CHU of Besançon (Dr. Doussot Alexandre).
Related publications:
- M. Privat , A. Massot , F. Hermetet, H. Al Sabea, C. Racoeur, N. Mabrouk , M. Cordonnier, M. Moreau, B. Collin, A. Bettaieb, F. Denat, E. Bodio, PS Bellaye, C. Goze, C. Paul. Development of an Immuno-SPECT/Fluorescent Bimodal Tracer Targeting Human or Murine PD-L1 on Preclinical Models. J Med Chem 2024.
- Privat M, Bellaye PS, Chazeau E, Racoeur C, Adumeau P, Vivier D, Bernhard C, Moreau M, Collin B, Bettaieb A, Denat F, Bodio E, Paul C, Goze C. First Comparison Study of the In Vitro and In Vivo Properties of a Randomly and Site-Specifically Conjugated SPECT/NIRF Monomolecular Multimodal Imaging Probe (MOMIP) Based on an aza-BODIPY Fluorophore. Bioconjug Chem 2023.
- M. Privat, P. S. Bellaye, R. Lescure, A. Massot, O. Baffroy, M. Moreau, C. Racoeur, G. Marcion, F. Denat, A. Bettaieb, B. Collin, E. Bodio, C. Paul, C. Goze, Development of an Easily Bioconjugatable Water-Soluble Single-Photon Emission-Computed Tomography/Optical Imaging Bimodal Imaging Probe Based on the aza-BODIPY Fluorophore. J Med Chem, (2021).
Hematology – lymphoma / myeloma
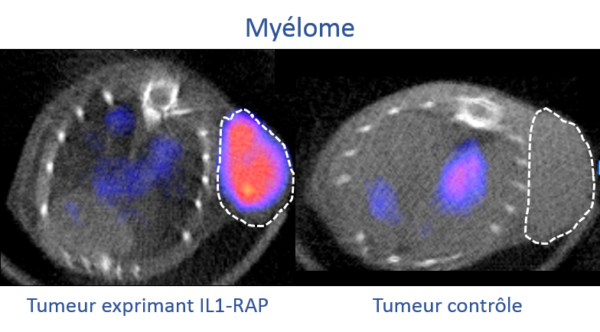
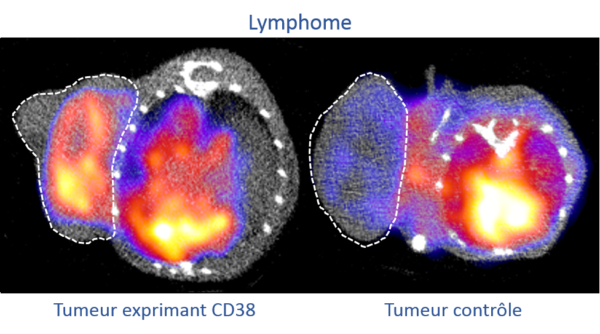
Our hematology projects aim to develop new imaging agents to better detect these cancers and offer solutions that facilitate patient monitoring and therapeutic decisions. Our research focuses on two main targets: the IL-1RAP protein, involved in inflammation and tumor development, is found overexpressed in several cancers, including acute and chronic myeloid leukemias. And the CD38 receptor, currently used clinically in the treatment of myeloma, anti-CD38 antibodies (e.g., Daratumumab) could also prove effective in certain forms of lymphoma, such as Burkitt lymphoma, which mainly affects children and young adults.
Hematological cancers or “blood cancers” affect approximately 35,000 people each year in France and mainly concern children, young adults and the elderly. Leukemia, lymphoma, and myeloma are the most common. Hematological cancers occur when the maturation of blood cells—white blood cells, red blood cells, and platelets—is impaired, which promotes their abnormal proliferation. Blood cells first develop in the bone marrow, deep within the bones, before being released into the blood. The accumulation of altered blood cells hinders the normal functioning of other cells (fighting infections, preventing bleeding, etc.). The earlier the alterations appear in the maturation process, the more aggressive the cancer is likely to be. The discovery of new early biomarkers of progression and treatment efficacy in this type of pathology is crucial for improving patient care.
In this context, we are working in collaboration with Dr. Christophe Ferrand ( CanCell Therapeutics , Besançon) to develop a companion imaging biomarker for a new CAR-T cell therapy targeting IL-1RAP. This innovative therapy is currently in clinical trials (Besançon University Hospital, CanCell Therapeutics), and a companion imaging biomarker targeting IL-1RAP could help better select patients with overexpression of this target and for whom this therapy is most likely to work. Our goal is therefore to develop a diagnostic companion based on an antibody targeting IL-1RAP modified to visualize it using medical imaging by incorporating a radioactive isotope visible on PET/CT, SPECT/CT, and/or PET/MRI imaging. Using this technology, it is also possible to replace the radioactive imaging isotope with another, more irradiating isotope, thus making it possible to obtain a therapeutic agent using the vectorized internal radiotherapy technique in order to specifically eliminate tumor cells.
To demonstrate the usefulness of targeting the CD38 receptor in lymphomas, we are currently testing anti-CD38 antibodies in different lymphoma models in collaboration with the hematology department of the Dijon University Hospital. To do this, we are slightly modifying these antibodies so that they can be observed using medical imaging techniques (PET/CT, SPECT/CT, PET/MRI). In this context, it is also possible to replace the radioactive imaging isotope with another, more irradiating isotope, thus making it possible to obtain a therapeutic agent via the vectorized internal radiotherapy technique in order to specifically eliminate tumor cells.
Imaging of cell therapies (e.g. CAR-T cells)
Cell therapies are innovative therapies in several pathologies including cancers, heart disease and autoimmune diseases. Cell therapy consists of either producing cells from a healthy donor in a laboratory or taking cells from a patient, then possibly modifying them to assign them a specific function, to reimplant them in the patient. These therapies often show strong efficacy in certain patients but are sometimes accompanied by adverse effects that can be serious. It is in this context that our research projects aim to give these therapeutic cells imaging capabilities in order to be able to monitor them in real time once reinjected into the patient. To this end, we propose modifying the cells so that they express on their surface proteins capable of trapping radioactive molecules for nuclear imaging. In this way, we will be able to monitor the accumulation of cells in tumors to evaluate their effectiveness and, at the same time, monitor the accumulation of cells in other organs to evaluate potential side effects. In this context we work in collaboration with Dr Alvaro BAEZA GARCIA (INSERM U1231, HSP-pathies team, Dr Carmen Garrido).
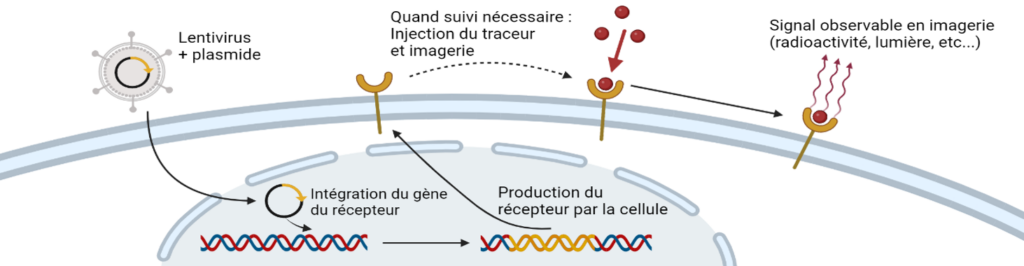
IDIOPATHIC PULMONARY FIBROSIS
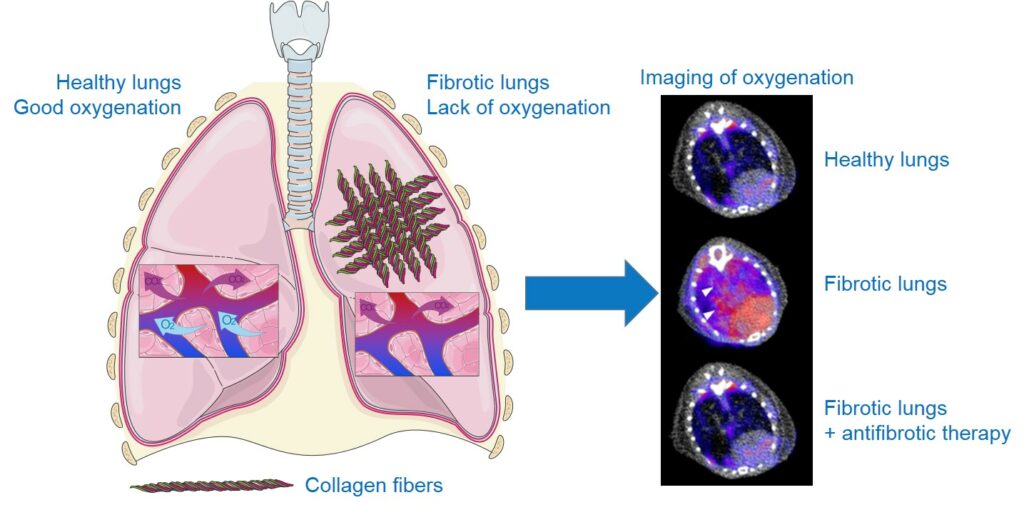
Idiopathic pulmonary fibrosis (IPF) is a rare, irreversible disease of unknown causes. It is a chronic condition characterized by progressive scarring of the lungs, leading to an accumulation of collagen-producing cells responsible for stiffening the lungs. As a result, the passage of oxygen and the release of carbon dioxide are no longer ensured, and patients primarily suffer from significant breathing difficulties such as progressive shortness of breath, both during exercise and at rest, a chronic dry cough, and severe fatigue. IPF generally begins after the age of 50 and has a rather poor prognosis, with a median survival of approximately 5 years after diagnosis. France is estimated to have at least 12,000 people affected and at least 4,400 new cases each year. Treatments remain very limited, with two compounds, pirfenidone and nintedanib, which have proven effective in reducing disease progression without completely halting it. Diagnostic difficulties and the lack of tools for monitoring disease progression and/or treatment effectiveness are significant clinical issues. Our goal is to find nuclear imaging tools that facilitate early diagnosis and patient monitoring in order to improve their care and survival.
Our platform has obtained €240,000 in ANR (HYMAGE-IPF) funding over three years (2020-2023) to develop, in collaboration with the INSERM HSP-pathies team led by Dr. Carmen Garrido and the Dijon University Hospital’s Rare Lung Diseases Reference Center led by Professor Philippe Bonniaud, imaging tracers for IPF targeting:
Hypoxia
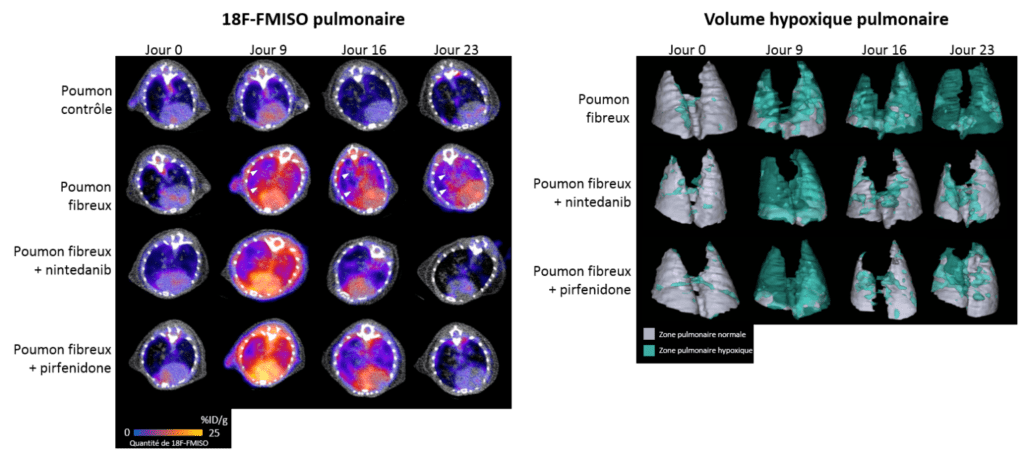
Due to the destruction of lung structure by excess scar tissue that disrupts gas exchange, hypoxia (lack of oxygen) is a significant characteristic of the lungs of patients with IPF. Using a radiotracer specific to hypoxic regions, 18F-FMISO (a radioactive molecule that specifically accumulates in cells under oxygen deficiency), we were able to show that areas of hypoxia were more prominent in lungs with fibrosis, correlating with disease severity. Furthermore, the level of pulmonary hypoxia measured by imaging with 18F-FMISO is a good predictive marker of disease progression and response to the anti-fibrosis treatments nintedanib and pirfenidone in our preclinical models (for more details, see the associated publication).
Related publication:
- Tanguy, F. Goirand, A. Bouchard, J. Frenay, M. Moreau, C. Mothes, A. Oudot, A. Helbling, M. Guillemin, P. Bonniaud, A. Cochet, B. Collin, P. S. Bellaye, [18F]FMISO PET/CT imaging of hypoxia as a non-invasive biomarker of disease progression and therapy efficacy in a preclinical model of pulmonary fibrosis: comparison with the [18F]FDG PET/CT approach. Eur J Nucl Med Mol Imaging, (2021).
- Bellaye PS, Beltramo G, Burgy O, Collin B, Cochet A, Bonniaud P. Measurement of hypoxia in the lung in idiopathic pulmonary fibrosis: a matter of control. Eur Respir J. 2022 Jan 27:2102711. doi: 10.1183/13993003.02711-2021.
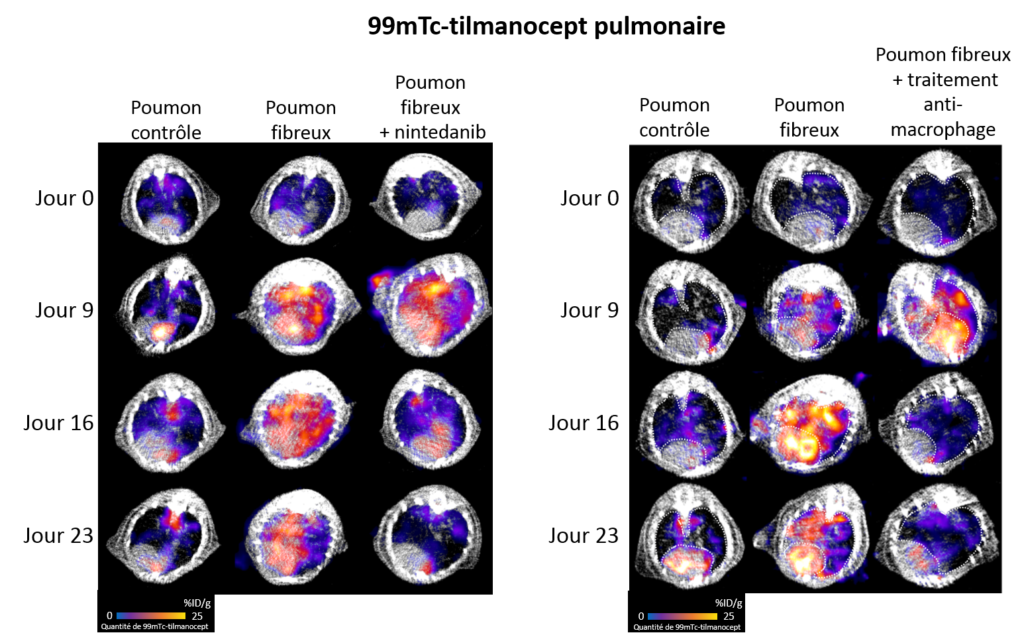
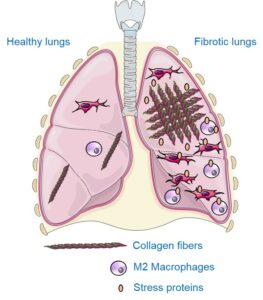
Macrophages
Macrophages are circulating and/or resident immune cells in various organs, including the lungs, where they provide part of the defenses against infections. In a context of fibrosis, these macrophages take on a particular phenotype, called M2 macrophage, and secrete growth factors, notably TGF-β, which promote the production of scar tissue including collagen. Thanks to the use of a radiotracer specific to M2 macrophages, 99mTc-tilmanocept (a radioactive molecule that binds specifically to M2 macrophages), we were able to show that M2 macrophages were more present in the lungs in conditions of fibrosis. In addition, the rate of pulmonary M2 macrophages measured by imaging with 99mTc-tilmanocept makes it possible to precisely monitor the progression of the disease and the response to the anti-fibrosis treatment nintedanib. Finally, the inhibition of these aggressive pro-fibrosis macrophages makes it possible to reduce the progression of pulmonary fibrosis in our preclinical models.
Related publication:
Heat shock proteins
Heat shock proteins (or stress proteins, HSPs) are proteins that are overexpressed under stress conditions to ensure cell survival. In certain conditions, including pulmonary fibrosis, these proteins promote disease progression. Our team has already shown that HSPs are involved in the overproduction of scar tissue under fibrosis conditions and that their inhibition is a promising strategy to combat this disease. Thus, they are interesting targets for developing imaging agents to visualize the progression of fibrosis and the response to treatments. Our team focuses on HSP90 and Gp96 proteins, which are overexpressed and/or secreted in high quantities in patients with IPF and in preclinical models. Specific radioactive probes for imaging HSP90 and Gp96 in fibrosis are currently being developed at IMATHERA.
Related post:
- J Tanguy , PM Boutanquoi, O Burgy, L Dondaine, G Beltramo, B Uyanik, C Garrido, P Bonniaud, PS Bellaye, F Goirand. HSPB5 Inhibition by NCI-41356 Reduces Experimental Lung Fibrosis by Blocking TGF-β1 Signaling. Pharmaceuticals (Basel). 2023.
- P. S. Bellaye, O. Burgy, P. Bonniaud, M. Kolb, HSP47: a potential target for fibrotic diseases and implications for therapy. Expert Opin Ther Targets 25, 49-62 (2021).
CARDIOLOGY
Early detection of cardiac effects of anticancer drugs
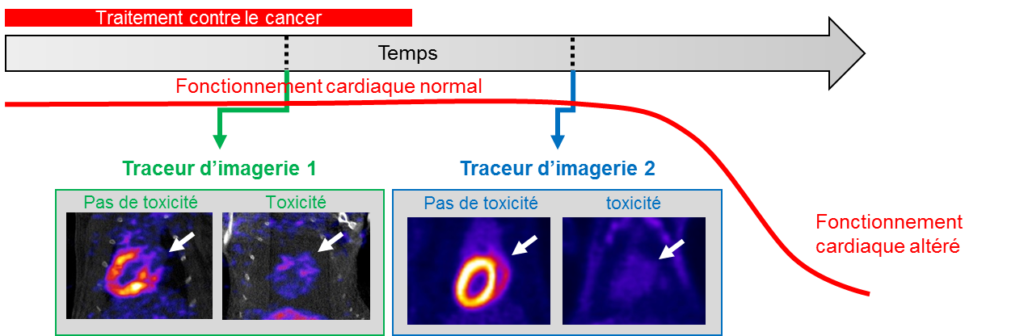
Over the past 20 years, the prognosis for cancer patients has steadily improved. However, the potential long-term complications of certain treatments, particularly cardiac complications, are now a major concern.
Indeed, cardiac complications of cancer treatments, although rare, are currently treated late when the heart muscle becomes less efficient. The objective of the work carried out in the laboratory is to develop imaging tracers (molecules injected into patients and then detected by non-invasive imaging) to detect the cardiac impact of treatments before it affects the heart pump.
Related publications:
METHODOLOGY IN PRECLINICAL IMAGING
Medical physics and instrumentation play an important role in our research program. This aspect of our activity is organized into three main areas.
Instrumentation
- Development of innovative imaging systems in collaboration with our academic and industrial partners. During IMAPPI, we initiated collaborations with Bioscan, Trifoil Imaging, and MR Solutions Ltd.
- Calibration and fine-tuning of imagers, in order to obtain high-resolution morphological images (CT, MRI) aligned with quantitative functional data (SPECT, PET).
- Reduced interference between PET and MRI systems.
- Performance evaluation of imagers based on international standards.
- Design of quality assurance protocols.
- Technological monitoring in order to maintain a high level of performance and adaptability to scientific projects.
Imaging of the living
- Design of morphological, quantitative, and functional imaging procedures to meet the needs of fundamental or applied research teams.
- Participation in the in vivo validation of the use of molecular probes and nanomaterials, compatible in particular with PET/MRI.
Dosimetry of ionizing radiation
- Evaluation of the absorbed dose resulting from nuclear or radiological examinations.
- Optimization of quantitative data analysis methods.
- Implementation of personalized dosimetry of vectorized internal radiotherapy.
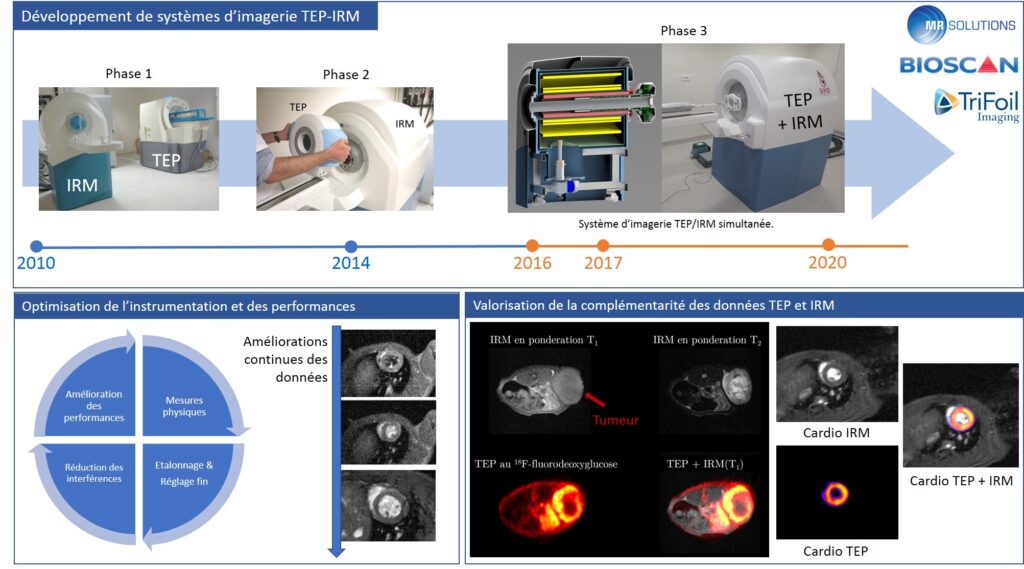
Since 2012, the preclinical imaging platform has hosted the Equipex “investments for the future” IMAPPI (Integrated Magnetic Resonance and Positron Emission Tomography in Preclinical Imaging) program, funded by the French National Agency for Research (ANR) to the tune of €7.3 million. During the three development phases of the IMAPPI project, a preclinical PET/MRI imaging prototype, in which the PET scanner is integrated into the center of the MRI magnet, was designed, assembled, and validated. This system, whose performance was published in 2021, has since been commercialized and is being implemented in several research teams around the world.
Starting in 2021, the IMAPPI project is entering a new research phase, which aims to take advantage of the high level of complementarity between PET and MRI data. Our current projects focus, among other things, on the following areas:
- improving cardiac imaging, particularly for the early detection of cardiotoxic effects of anticancer chemotherapy and imaging of ischemic heart disease.
- imaging of the tumor microenvironment and biomarkers of interest in immunotherapy, in particular microcirculation and tissue oxygenation.
- the integration of radiomics and artificial intelligence into projects.
Related publications:
- Courteau, A., J. McGrath, P. M. Walker, B. Presles, R. Garipov, A. Cochet, F. Brunotte, and J. M. Vrigneaud. A Practical Quality Assurance Procedure for Data Acquisitions in Preclinical Simultaneous Pet/Mr Systems. Mol Imaging Biol 25 2023, 450-63
- Courteau A, McGrath J, Walker PM, Pegg R, Martin G, Garipov R, Doughty P, Cochet A, Brunotte F, Vrigneaud JM. Performance Evaluation and Compatibility Studies of a Compact Preclinical Scanner for Simultaneous PET/MR Imaging at 7 Tesla. 2021. IEEE Trans Med Imaging, 40(1):205-217.
- Vrigneaud J.M., McGrath J., Courteau A., Pegg R., Gomis A.S.-P., Camacho A., Martin G., Schramm N., Brunotte F. Initial performance evaluation of a preclinical PET scanner available as a clip-on assembly in a sequential PET/MRI system. 2018. Phys. Med. Biol. 63, 125007.
- S. Bricq, H. L. Kidane, J. Zavala-Bojorquez, A. Oudot, J. M. Vrigneaud, F. Brunotte, P. M. Walker, A. Cochet, A. Lalande, Automatic deformable PET/MRI registration for preclinical studies based on B-splines and non-linear intensity transformation. Med Biol Eng Comput 56, 1531-1539 (2018).
- Vrigneaud J.M., Walker P., Barbier B., Camacho A., Oudot A., Collin B. Brunotte F. Performance evaluation of the PET component of a sequential APD-based micro-PET/MR imaging system. 2017. Biomed. Phys. Eng. Express 3, 035006.
- Pierre-Marc Jodoin, Fredy Pinheiro, Alexandra Oudot, Alain Lalande. Left-Ventricle Segmentation of SPECT Images of Rats. 2015. IEEE Transactions on Biomedical Engineering, Institute of Electrical and Electronics Engineers, 62(9), pp. 2260–2268.
- Brunotte F., Haas H., Collin B., Oudot A., Bricq S., Lalande A., Tizon X., Vrigneaud J.M., Walker P. Integrated PET/MRI in preclinical studies State of the art. Tijdschrift voor nucleaire geneeskunde, 2013, 35
- Vrigneaud JM, Courteau A, Ranouil J, Morgand L, Raguin O, Walker P, Oudot A, Collin B, Brunotte F. Application of the optically stimulated luminescence (OSL) technique for mouse dosimetry in micro-CT imaging. 2013. Med Phys.;40(12):122102.
OTHER PROJECTS AND PARTNERS
IMATHERA is also involved in many other projects in collaboration with other local or national research teams.
The main projects are:
Development of an imaging agent targeting CD8+ (cytotoxic) T lymphocytes to visualize the anti-tumor immune response: the BIOCAIR project. This project is funded by the ERDF.

Imaging of PDL1 protein (immunotherapy target) in colon cancer.
Development of theranostic agents combining the potential of fluorescence imaging, nuclear imaging, and photodynamic therapy (PDT). This project is funded by SATT-SAYENS.
Our main partners:

OTHER PUBLICATIONS
- Aleksandra Georgievski, Pierre-Simon Bellaye, Benjamin Tournier, Hélène Choubley, Jean-Paul Pais de Barros, Michaële Herbst, Arnaud Béduneau, Patrick Callier, Bertrand Collin, Frédérique Végran, Paola Ballerini, Carmen Garrido, Ronan Quéré. Valrubicin-loaded immunoliposomes for specific vesicle-mediated cell death in the treatment of hematological cancers. Cell Death Dis 2024.
- Kévin Guillen, Pierre-Olivier Comby, Alexandra Oudot, Anne-Virginie Salsac, Nicolas Falvo, Thierry Virely, Olivia Poupardin, Mélanie Guillemin, Olivier Chevallier, Romaric Loffroy. Iodixanol as a New Contrast Agent for Cyanoacrylate Embolization: A Preliminary In Vivo Swine Study. Biomedicines 2023.
- Vivian Lioret, Pierre-Simon Bellaye, Yann Bernhard, Mathieu Moreau, Mélanie Guillemin, Camille Drouet, Bertrand Collin, Richard Decréau. Cherenkov Radiation induced photodynamic therapy – repurposing older photosensitizers, and radionuclides. Photodiagnosis Photodyn Ther 2023.
- Marques, C., M. J. Hajipour, C. Marets, A. Oudot, R. Safavi-Sohi, M. Guillemin, G. Borchard, et al. Identification of the Proteins Determining the Blood Circulation Time of Nanoparticles. ACS Nano 17 2023, 12458-70.
- Nazaryan, S., A. Bruguière, N. Hovhannisyan, T. Miyamoto, A. M. M. Dias, P. S. Bellaye, B. Collin, L. Briand, and C. Mitaine-Offer. Oleanolic Acid Glycosides from Scabiosa Caucasica and Scabiosa Ochroleuca : Structural Analysis and Cytotoxicity. Molecules 28 2023.
- Vizier R, Garnier AR, Dias A, Moreau M, Claron M, Collin B, Denat F, Bellaye PS*, Goncalves V*. SPECT Imaging of Lysyl Oxidase-like 2 in a Model of Idiopathic Pulmonary Fibrosis. Mol Pharm 2023, 20:3613-22.
- Julie Tanguy, Pierre-Marie Boutanquoi, Olivier Burgy, Lucile Dondaine, Guillaume Beltramo, Burhan Uyanik, Carmen Garrido, Philippe Bonniaud, Pierre-Simon Bellaye and Françoise Goirand, HSPB5 Inhibition by NCI-41356 Reduces Experimental Lung Fibrosis by Blocking TGF-β1 Signaling. Pharmaceuticals 2023, 16, 177.
- Jame Frenay, Pierre-Simon Bellaye, Alexandra Oudot, Alex Helbling, Camille Petitot, Christophe Ferrand, Bertrand Collin, Alexandre M. M.Dias, IL-1RAP, a Key Therapeutic Target in Cancer, Int. J. Mol. Sci. 2022, 23(23), 14918.
- Dias AMM, Courteau A, Bellaye PS, Kohli E, Oudot A, Doulain PE, Petitot C, Walker PM, Decréau R, Collin B. Superparamagnetic Iron Oxide Nanoparticles for Immunotherapy of Cancers through Macrophages and Magnetic Hyperthermia. Pharmaceutics. 2022 Nov 5;14(11):2388.
- Baurand PE, Balland J, Reynas C, Ramseyer M, Vivier D, Bellaye PS, Collin B, Paul C, Denat F, Asgarov K, Pallandre JR, Ringenbach L. Development of Anti-LRRC15 Small Fragments for Imaging Purposes Using a Phage-Display ScFv Approach. Int J Mol Sci. 2022 Oct 21;23(20):12677.
- Pertuit D, Mitaine-Offer AC, Miyamoto T, Tanaka C, Delaude C, Bellaye PS, Collin B, Lacaille-Dubois MA. Oleanane-type glycosides isolated from the trunk barks of the Central African tree Millettia laurentii. Fitoterapia. 2022 Jun;159:105193.
- Limagne E, Nuttin L, Thibaudin M, Jacquin E, Aucagne R, Bon M, Revy S, Barnestein R, Ballot E, Truntzer C, Derangère V, Fumet JD, Latour C, Rébé C, Bellaye PS, Kaderbhaï CG, Spill A, Collin B, Callanan MB, Lagrange A, Favier L, Coudert B, Arnould L, Ladoire S, Routy B, Joubert P, Ghiringhelli F. MEK inhibition overcomes chemoimmunotherapy resistance by inducing CXCL10 in cancer cells. Cancer Cell. 2022 Jan 17:S1535-6108(21)00662-0. doi: 10.1016/j.ccell.2021.12.009.
- Nguyen DH, Mitaine-Offer AC, Miyamoto T, Tanaka C, Bellaye PS, Collin B, Chambin O, Lacaille-Dubois MA. Steroidal glycosides from the Vietnamese cultivar Cordyline fruticosa “Fairchild red”. Phytochemistry. 2021 Dec;192:112966.
- Bouchard A, Collin B, Garrido C, Bellaye PS, Kohli E. GARP: A Key Target to Evaluate Tumor Immunosuppressive Microenvironment. Biology (Basel). 2021 Aug 27;10(9):836
- E. Renard, M. Moreau, P. S. Bellaye, M. Guillemin, B. Collin, A. Prignon, F. Denat, V. Goncalves, Positron Emission Tomography Imaging of Neurotensin Receptor-Positive Tumors with 68Ga-Labeled Antagonists: The Chelate Makes the Difference Again. J Med Chem 64, 8564-8578 (2021).
- R. Mshaik, J. Simonet, A. Georgievski, L. Jamal, S. Bechoua, P. Ballerini, P. S. Bellaye, Z. Mlamla, J. P. Pais de Barros, A. Geissler, P. J. Francin, F. Girodon, C. Garrido, R. Quéré, HSP90 inhibitor NVP-BEP800 affects stability of SRC kinases and growth of T-cell and B-cell acute lymphoblastic leukemias. Blood Cancer J 11, 61 (2021).
- Marcion, F. Hermetet, F. Neiers, B. Uyanik, L. Dondaine, A. M. M. Dias, L. Da Costa, M. Moreau, P. S. Bellaye, B. Collin, J. Gobbo, L. Briand, R. Seigneuric, O. Kitten, M. Cinier, C. Garrido, Nanofitins targeting heat shock protein 110: An innovative immunotherapeutic modality in cancer. Int J Cancer 148, 3019-3031 (2021).
- Lescure, M. Privat, J. Pliquett, A. Massot, O. Baffroy, B. Busser, P. S. Bellaye, B. Collin, F. Denat, A. Bettaïeb, L. Sancey, C. Paul, C. Goze, E. Bodio, Near-infrared emitting fluorescent homobimetallic gold(I) complexes displaying promising in vitro and in vivo therapeutic properties. Eur J Med Chem 220, 113483 (2021).
- V. Lioret, P. S. Bellaye, C. Arnould, B. Collin, R. A. Decréau, Dual Cherenkov Radiation-Induced Near-Infrared Luminescence Imaging and Photodynamic Therapy toward Tumor Resection. J Med Chem 63, 9446-9456 (2020).
- Hadi, C. Ramseyer, T. Gautier, P. S. Bellaye, T. Lopez, A. Schmitt, S. Foley, S. Yesylevskyy, T. Minervini, R. Douhard, L. Dondaine, L. Proukhnitzky, S. Messaoudi, M. Wendremaire, M. Moreau, F. Neiers, B. Collin, F. Denat, L. Lagrost, C. Garrido, F. Lirussi, Lipoproteins LDL versus HDL as nanocarriers to target either cancer cells or macrophages. JCI Insight 5, (2020).
- O. Florès, J. Pliquett, L. Abad Galan, R. Lescure, F. Denat, O. Maury, A. Pallier, P. S. Bellaye, B. Collin, S. Même, C. S. Bonnet, E. Bodio, C. Goze, Aza-BODIPY Platform: Toward an Efficient Water-Soluble Bimodal Imaging Probe for MRI and Near-Infrared Fluorescence. Inorg Chem 59, 1306-1314 (2020).
- Thomas, J. Boudon, L. Maurizi, M. Moreau, P. Walker, I. Severin, A. Oudot, C. Goze, S. Poty, J. M. Vrigneaud, F. Demoisson, F. Denat, F. Brunotte, N. Millot, Innovative Magnetic Nanoparticles for PET/MRI Bimodal Imaging. ACS Omega 4, 2637-2648 (2019).
- J. Pliquett, A. Dubois, C. Racoeur, N. Mabrouk, S. Amor, R. Lescure, A. Bettaïeb, B. Collin, C. Bernhard, F. Denat, P. S. Bellaye, C. Paul, E. Bodio, C. Goze, A Promising Family of Fluorescent Water-Soluble aza-BODIPY Dyes for in Vivo Molecular Imaging. Bioconjug Chem 30, 1061-1066 (2019).
- D. H. Nguyen, A. C. Mitaine-Offer, S. Maroso, A. M. Papini, T. Paululat, P. S. Bellaye, B. Collin, O. Chambin, M. A. Lacaille-Dubois, Cytotoxic glycosides from the roots of Weigela x \”Bristol Ruby\”. Fitoterapia 137, 104242 (2019).
- A. Loiseau, J. Boudon, A. Oudot, M. Moreau, R. Boidot, R. Chassagnon, N. M. Saïd, S. Roux, C. Mirjolet, N. Millot, Titanate Nanotubes Engineered with Gold Nanoparticles and Docetaxel to Enhance Radiotherapy on Xenografted Prostate Tumors. Cancers (Basel) 11, (2019).
- G. Leterme, C. Guigou, A. Oudot, B. Collin, J. Boudon, N. Millot, A. Geissler, K. Belharet, A. Bozorg Grayeli, Superparamagnetic Nanoparticle Delivery to the Cochlea Through Round Window by External Magnetic Field: Feasibility and Toxicity. Surg Innov 26, 646-655 (2019).
- C. Canovas, M. Moreau, J. M. Vrigneaud, P. S. Bellaye, B. Collin, F. Denat, V. Goncalves, Modular Assembly of Multimodal Imaging Agents through an Inverse Electron Demand Diels-Alder Reaction. Bioconjug Chem 30, 888-897 (2019).
- A. Bernard, S. Chevrier, F. Beltjens, M. Dosset, E. Viltard, A. Lagrange, V. Derangère, A. Oudot, F. Ghiringhelli, B. Collin, L. Apetoh, O. Feron, S. Chen, L. Arnould, F. Végran, R. Boidot, Cleaved Caspase-3 Transcriptionally Regulates Angiogenesis-Promoting Chemotherapy Resistance. Cancer Res 79, 5958-5970 (2019).
- V. L. Tran, V. Thakare, M. Natuzzi, M. Moreau, A. Oudot, J. M. Vrigneaud, A. Courteau, C. Louis, S. Roux, F. Boschetti, F. Denat, O. Tillement, F. Lux, Functionalization of Gadolinium Chelates Silica Nanoparticle through Silane Chemistry for Simultaneous MRI/ 64Cu PET Imaging. Contrast Media Mol Imaging 2018, 7938267 (2018).
- E. Desvaux, A. Courteau, P. S. Bellaye, M. Guillemin, C. Drouet, P. Walker, B. Collin, R. A. Decréau, Cherenkov luminescence imaging is a fast and relevant preclinical tool to assess tumour hypoxia in vivo. EJNMMI Res 8, 111 (2018).
- C. Canovas, M. Moreau, C. Bernhard, A. Oudot, M. Guillemin, F. Denat, V. Goncalves, Site-Specific Dual Labeling of Proteins on Cysteine Residues with Chlorotetrazines. Angew Chem Int Ed Engl 57, 10646-10650 (2018).
- C. Canovas, P. S. Bellaye, M. Moreau, A. Romieu, F. Denat, V. Goncalves, Site-specific near-infrared fluorescent labelling of proteins on cysteine residues with meso-chloro-substituted heptamethine cyanine dyes. Org Biomol Chem 16, 8831-8836 (2018).
- P. S. Bellaye, A. Oudot, J. M. Vrigneaud, O. Raguin, F. Bichat, A. Vaslin, H. Maby-El Hajjami, C. Zanna, G. Vuagniaux, P. Fumoleau, F. Denat, F. Brunotte, B. Collin, Nuclear Imaging Study of the Pharmacodynamic Effects of Debio 1143, an Antagonist of Multiple Inhibitor of Apoptosis Proteins (IAPs), in a Triple-Negative Breast Cancer Model. Contrast Media Mol Imaging 2018, 8494031 (2018).
- P. S. Bellaye, M. Moreau, O. Raguin, A. Oudot, C. Bernhard, J. M. Vrigneaud, L. Dumont, D. Vandroux, F. Denat, A. Cochet, F. Brunotte, B. Collin, Radiolabeled F(ab”) <sub>2</sub->cetuximab for theranostic purposes in colorectal and skin tumor-bearing mice models. Clin Transl Oncol 20, 1557-1570 (2018).
- M. Moreau, S. Poty, J. M. Vrigneaud, P. Walker, M. Guillemin, O. Raguin, A. Oudot, C. Bernhard, C. Goze, F. Boschetti, B. Collin, F. Brunotte, F. Denat, MANOTA: a promising bifunctional chelating agent for copper-64 immunoPET. Dalton Trans 46, 14659-14668 (2017).
- C. Mirjolet, J. Boudon, A. Loiseau, S. Chevrier, R. Boidot, A. Oudot, B. Collin, E. Martin, P. A. Joy, N. Millot, G. Créhange, Docetaxel-titanate nanotubes enhance radiosensitivity in an androgen-independent prostate cancer model. Int J Nanomedicine 12, 6357-6364 (2017).
- Y. Bernhard, B. Collin, R. A. Decréau, Redshifted Cherenkov Radiation for in vivo Imaging: Coupling Cherenkov Radiation Energy Transfer to multiple Förster Resonance Energy Transfers. Sci Rep 7, 45063 (2017).
- N. Maindron, M. Ipuy, C. Bernhard, D. Lhenry, M. Moreau, S. Carme, A. Oudot, B. Collin, J. M. Vrigneaud, P. Provent, F. Brunotte, F. Denat, C. Goze, Near-Infrared-Emitting BODIPY-trisDOTA(111) In as a Monomolecular Multifunctional Imaging Probe: From Synthesis to In Vivo Investigations. Chemistry 22, 12670-12674 (2016).
- Laurent, C. Bernhard, S. Dufort, G. Jiménez Sánchez, R. Bazzi, F. Boschetti, M. Moreau, T. H. Vu, B. Collin, A. Oudot, N. Herath, H. Requardt, S. Laurent, L. Vander Elst, R. Muller, M. Dutreix, M. Meyer, F. Brunotte, P. Perriat, F. Lux, O. Tillement, G. Le Duc, F. Denat, S. Roux, Minor changes in the macrocyclic ligands but major consequences on the efficiency of gold nanoparticles designed for radiosensitization. Nanoscale 8, 12054-12065 (2016).
- D. Lhenry, M. Larrouy, C. Bernhard, V. Goncalves, O. Raguin, P. Provent, M. Moreau, B. Collin, A. Oudot, J. M. Vrigneaud, F. Brunotte, C. Goze, F. Denat, BODIPY: A Highly Versatile Platform for the Design of Bimodal Imaging Probes. Chemistry 21, 13091-13099 (2015).
- M. Jodoin, F. Pinheiro, A. Oudot, A. Lalande, Left-Ventricle Segmentation of SPECT Images of Rats. IEEE Trans Biomed Eng 62, 2260-2268 (2015).
- Duheron, M. Moreau, B. Collin, W. Sali, C. Bernhard, C. Goze, T. Gautier, J. P. Pais de Barros, V. Deckert, F. Brunotte, L. Lagrost, F. Denat, Dual labeling of lipopolysaccharides for SPECT-CT imaging and fluorescence microscopy. ACS Chem Biol 9, 656-662 (2014).
- De Taddéo, B. Collin, H. Hardy, B. Guichard, P. Trouilloud, O. Trost, [Can one harvest a long bone stick in the radial forearm flap? Original radioanatomical and NanoSPECT-CT Bioscan microvascular study]. Ann Chir Plast Esthet 59, 170-176 (2014).
- Y. Bernhard, B. Collin, R. A. Decréau, Inter/intramolecular Cherenkov radiation energy transfer (CRET) from a fluorophore with a built-in radionuclide. Chem Commun (Camb) 50, 6711-6713 (2014).
- J. M. Vrigneaud, A. Courteau, J. Ranouil, L. Morgand, O. Raguin, P. Walker, A. Oudot, B. Collin, F. Brunotte, Application of the optically stimulated luminescence (OSL) technique for mouse dosimetry in micro-CT imaging. Med Phys 40, 122102 (2013).
- F. Végran, R. Mary, A. Gibeaud, C. Mirjolet, B. Collin, A. Oudot, C. Charon-Barra, L. Arnould, S. Lizard-Nacol, R. Boidot, Survivin-3B potentiates immune escape in cancer but also inhibits the toxicity of cancer chemotherapy. Cancer Res 73, 5391-5401 (2013).
- Moreau, O. Raguin, J. M. Vrigneaud, B. Collin, C. Bernhard, X. Tizon, F. Boschetti, O. Duchamp, F. Brunotte, F. Denat, DOTAGA-trastuzumab. A new antibody conjugate targeting HER2/Neu antigen for diagnostic purposes. Bioconjug Chem 23, 1181-1188 (2012).
- * Crédit photo mise en avant: A. Chezières
News from the Platform
Other publications
- Aleksandra Georgievski, Pierre-Simon Bellaye, Benjamin Tournier, Hélène Choubley, Jean-Paul Pais de Barros, Michaële Herbst, Arnaud Béduneau, Patrick Callier, Bertrand Collin, Frédérique Végran, Paola Ballerini, Carmen Garrido, Ronan Quéré. Valrubicin-loaded immunoliposomes for specific vesicle-mediated cell death in the treatment of hematological cancers. Cell Death Dis 2024.
- Kévin Guillen, Pierre-Olivier Comby, Alexandra Oudot, Anne-Virginie Salsac, Nicolas Falvo, Thierry Virely, Olivia Poupardin, Mélanie Guillemin, Olivier Chevallier, Romaric Loffroy. Iodixanol as a New Contrast Agent for Cyanoacrylate Embolization: A Preliminary In Vivo Swine Study. Biomedicines 2023.
- Vivian Lioret, Pierre-Simon Bellaye, Yann Bernhard, Mathieu Moreau, Mélanie Guillemin, Camille Drouet, Bertrand Collin, Richard Decréau. Cherenkov Radiation induced photodynamic therapy – repurposing older photosensitizers, and radionuclides. Photodiagnosis Photodyn Ther 2023.
- Marques, C., M. J. Hajipour, C. Marets, A. Oudot, R. Safavi-Sohi, M. Guillemin, G. Borchard, et al. Identification of the Proteins Determining the Blood Circulation Time of Nanoparticles. ACS Nano 17 2023, 12458-70.
- Nazaryan, S., A. Bruguière, N. Hovhannisyan, T. Miyamoto, A. M. M. Dias, P. S. Bellaye, B. Collin, L. Briand, and C. Mitaine-Offer. Oleanolic Acid Glycosides from Scabiosa Caucasica and Scabiosa Ochroleuca : Structural Analysis and Cytotoxicity. Molecules 28 2023.
- Vizier R, Garnier AR, Dias A, Moreau M, Claron M, Collin B, Denat F, Bellaye PS*, Goncalves V*. SPECT Imaging of Lysyl Oxidase-like 2 in a Model of Idiopathic Pulmonary Fibrosis. Mol Pharm 2023, 20:3613-22.
- Julie Tanguy, Pierre-Marie Boutanquoi, Olivier Burgy, Lucile Dondaine, Guillaume Beltramo, Burhan Uyanik, Carmen Garrido, Philippe Bonniaud, Pierre-Simon Bellaye and Françoise Goirand, HSPB5 Inhibition by NCI-41356 Reduces Experimental Lung Fibrosis by Blocking TGF-β1 Signaling. Pharmaceuticals 2023, 16, 177.
- Jame Frenay, Pierre-Simon Bellaye, Alexandra Oudot, Alex Helbling, Camille Petitot, Christophe Ferrand, Bertrand Collin, Alexandre M. M.Dias, IL-1RAP, a Key Therapeutic Target in Cancer, Int. J. Mol. Sci. 2022, 23(23), 14918.
- Dias AMM, Courteau A, Bellaye PS, Kohli E, Oudot A, Doulain PE, Petitot C, Walker PM, Decréau R, Collin B. Superparamagnetic Iron Oxide Nanoparticles for Immunotherapy of Cancers through Macrophages and Magnetic Hyperthermia. Pharmaceutics. 2022 Nov 5;14(11):2388.
- Baurand PE, Balland J, Reynas C, Ramseyer M, Vivier D, Bellaye PS, Collin B, Paul C, Denat F, Asgarov K, Pallandre JR, Ringenbach L. Development of Anti-LRRC15 Small Fragments for Imaging Purposes Using a Phage-Display ScFv Approach. Int J Mol Sci. 2022 Oct 21;23(20):12677.
- Pertuit D, Mitaine-Offer AC, Miyamoto T, Tanaka C, Delaude C, Bellaye PS, Collin B, Lacaille-Dubois MA. Oleanane-type glycosides isolated from the trunk barks of the Central African tree Millettia laurentii. Fitoterapia. 2022 Jun;159:105193.
- Limagne E, Nuttin L, Thibaudin M, Jacquin E, Aucagne R, Bon M, Revy S, Barnestein R, Ballot E, Truntzer C, Derangère V, Fumet JD, Latour C, Rébé C, Bellaye PS, Kaderbhaï CG, Spill A, Collin B, Callanan MB, Lagrange A, Favier L, Coudert B, Arnould L, Ladoire S, Routy B, Joubert P, Ghiringhelli F. MEK inhibition overcomes chemoimmunotherapy resistance by inducing CXCL10 in cancer cells. Cancer Cell. 2022 Jan 17:S1535-6108(21)00662-0. doi: 10.1016/j.ccell.2021.12.009.
- Nguyen DH, Mitaine-Offer AC, Miyamoto T, Tanaka C, Bellaye PS, Collin B, Chambin O, Lacaille-Dubois MA. Steroidal glycosides from the Vietnamese cultivar Cordyline fruticosa “Fairchild red”. Phytochemistry. 2021 Dec;192:112966.
- Bouchard A, Collin B, Garrido C, Bellaye PS, Kohli E. GARP: A Key Target to Evaluate Tumor Immunosuppressive Microenvironment. Biology (Basel). 2021 Aug 27;10(9):836
- E. Renard, M. Moreau, P. S. Bellaye, M. Guillemin, B. Collin, A. Prignon, F. Denat, V. Goncalves, Positron Emission Tomography Imaging of Neurotensin Receptor-Positive Tumors with 68Ga-Labeled Antagonists: The Chelate Makes the Difference Again. J Med Chem 64, 8564-8578 (2021).
- R. Mshaik, J. Simonet, A. Georgievski, L. Jamal, S. Bechoua, P. Ballerini, P. S. Bellaye, Z. Mlamla, J. P. Pais de Barros, A. Geissler, P. J. Francin, F. Girodon, C. Garrido, R. Quéré, HSP90 inhibitor NVP-BEP800 affects stability of SRC kinases and growth of T-cell and B-cell acute lymphoblastic leukemias. Blood Cancer J 11, 61 (2021).
- Marcion, F. Hermetet, F. Neiers, B. Uyanik, L. Dondaine, A. M. M. Dias, L. Da Costa, M. Moreau, P. S. Bellaye, B. Collin, J. Gobbo, L. Briand, R. Seigneuric, O. Kitten, M. Cinier, C. Garrido, Nanofitins targeting heat shock protein 110: An innovative immunotherapeutic modality in cancer. Int J Cancer 148, 3019-3031 (2021).
- Lescure, M. Privat, J. Pliquett, A. Massot, O. Baffroy, B. Busser, P. S. Bellaye, B. Collin, F. Denat, A. Bettaïeb, L. Sancey, C. Paul, C. Goze, E. Bodio, Near-infrared emitting fluorescent homobimetallic gold(I) complexes displaying promising in vitro and in vivo therapeutic properties. Eur J Med Chem 220, 113483 (2021).
- V. Lioret, P. S. Bellaye, C. Arnould, B. Collin, R. A. Decréau, Dual Cherenkov Radiation-Induced Near-Infrared Luminescence Imaging and Photodynamic Therapy toward Tumor Resection. J Med Chem 63, 9446-9456 (2020).
- Hadi, C. Ramseyer, T. Gautier, P. S. Bellaye, T. Lopez, A. Schmitt, S. Foley, S. Yesylevskyy, T. Minervini, R. Douhard, L. Dondaine, L. Proukhnitzky, S. Messaoudi, M. Wendremaire, M. Moreau, F. Neiers, B. Collin, F. Denat, L. Lagrost, C. Garrido, F. Lirussi, Lipoproteins LDL versus HDL as nanocarriers to target either cancer cells or macrophages. JCI Insight 5, (2020).
- O. Florès, J. Pliquett, L. Abad Galan, R. Lescure, F. Denat, O. Maury, A. Pallier, P. S. Bellaye, B. Collin, S. Même, C. S. Bonnet, E. Bodio, C. Goze, Aza-BODIPY Platform: Toward an Efficient Water-Soluble Bimodal Imaging Probe for MRI and Near-Infrared Fluorescence. Inorg Chem 59, 1306-1314 (2020).
- Thomas, J. Boudon, L. Maurizi, M. Moreau, P. Walker, I. Severin, A. Oudot, C. Goze, S. Poty, J. M. Vrigneaud, F. Demoisson, F. Denat, F. Brunotte, N. Millot, Innovative Magnetic Nanoparticles for PET/MRI Bimodal Imaging. ACS Omega 4, 2637-2648 (2019).
- J. Pliquett, A. Dubois, C. Racoeur, N. Mabrouk, S. Amor, R. Lescure, A. Bettaïeb, B. Collin, C. Bernhard, F. Denat, P. S. Bellaye, C. Paul, E. Bodio, C. Goze, A Promising Family of Fluorescent Water-Soluble aza-BODIPY Dyes for in Vivo Molecular Imaging. Bioconjug Chem 30, 1061-1066 (2019).
- D. H. Nguyen, A. C. Mitaine-Offer, S. Maroso, A. M. Papini, T. Paululat, P. S. Bellaye, B. Collin, O. Chambin, M. A. Lacaille-Dubois, Cytotoxic glycosides from the roots of Weigela x \”Bristol Ruby\”. Fitoterapia 137, 104242 (2019).
- A. Loiseau, J. Boudon, A. Oudot, M. Moreau, R. Boidot, R. Chassagnon, N. M. Saïd, S. Roux, C. Mirjolet, N. Millot, Titanate Nanotubes Engineered with Gold Nanoparticles and Docetaxel to Enhance Radiotherapy on Xenografted Prostate Tumors. Cancers (Basel) 11, (2019).
- G. Leterme, C. Guigou, A. Oudot, B. Collin, J. Boudon, N. Millot, A. Geissler, K. Belharet, A. Bozorg Grayeli, Superparamagnetic Nanoparticle Delivery to the Cochlea Through Round Window by External Magnetic Field: Feasibility and Toxicity. Surg Innov 26, 646-655 (2019).
- C. Canovas, M. Moreau, J. M. Vrigneaud, P. S. Bellaye, B. Collin, F. Denat, V. Goncalves, Modular Assembly of Multimodal Imaging Agents through an Inverse Electron Demand Diels-Alder Reaction. Bioconjug Chem 30, 888-897 (2019).
- A. Bernard, S. Chevrier, F. Beltjens, M. Dosset, E. Viltard, A. Lagrange, V. Derangère, A. Oudot, F. Ghiringhelli, B. Collin, L. Apetoh, O. Feron, S. Chen, L. Arnould, F. Végran, R. Boidot, Cleaved Caspase-3 Transcriptionally Regulates Angiogenesis-Promoting Chemotherapy Resistance. Cancer Res 79, 5958-5970 (2019).
- V. L. Tran, V. Thakare, M. Natuzzi, M. Moreau, A. Oudot, J. M. Vrigneaud, A. Courteau, C. Louis, S. Roux, F. Boschetti, F. Denat, O. Tillement, F. Lux, Functionalization of Gadolinium Chelates Silica Nanoparticle through Silane Chemistry for Simultaneous MRI/ <64>/<sup>Cu PET Imaging. Contrast Media Mol Imaging 2018, 7938267 (2018).
- E. Desvaux, A. Courteau, P. S. Bellaye, M. Guillemin, C. Drouet, P. Walker, B. Collin, R. A. Decréau, Cherenkov luminescence imaging is a fast and relevant preclinical tool to assess tumour hypoxia in vivo. EJNMMI Res 8, 111 (2018).
- C. Canovas, M. Moreau, C. Bernhard, A. Oudot, M. Guillemin, F. Denat, V. Goncalves, Site-Specific Dual Labeling of Proteins on Cysteine Residues with Chlorotetrazines. Angew Chem Int Ed Engl 57, 10646-10650 (2018).
- C. Canovas, P. S. Bellaye, M. Moreau, A. Romieu, F. Denat, V. Goncalves, Site-specific near-infrared fluorescent labelling of proteins on cysteine residues with meso-chloro-substituted heptamethine cyanine dyes. Org Biomol Chem 16, 8831-8836 (2018).
- P. S. Bellaye, A. Oudot, J. M. Vrigneaud, O. Raguin, F. Bichat, A. Vaslin, H. Maby-El Hajjami, C. Zanna, G. Vuagniaux, P. Fumoleau, F. Denat, F. Brunotte, B. Collin, Nuclear Imaging Study of the Pharmacodynamic Effects of Debio 1143, an Antagonist of Multiple Inhibitor of Apoptosis Proteins (IAPs), in a Triple-Negative Breast Cancer Model. Contrast Media Mol Imaging 2018, 8494031 (2018).
- P. S. Bellaye, M. Moreau, O. Raguin, A. Oudot, C. Bernhard, J. M. Vrigneaud, L. Dumont, D. Vandroux, F. Denat, A. Cochet, F. Brunotte, B. Collin, Radiolabeled F(ab”) <sub>2</sub->cetuximab for theranostic purposes in colorectal and skin tumor-bearing mice models. Clin Transl Oncol 20, 1557-1570 (2018).
- M. Moreau, S. Poty, J. M. Vrigneaud, P. Walker, M. Guillemin, O. Raguin, A. Oudot, C. Bernhard, C. Goze, F. Boschetti, B. Collin, F. Brunotte, F. Denat, MANOTA: a promising bifunctional chelating agent for copper-64 immunoPET. Dalton Trans 46, 14659-14668 (2017).
- C. Mirjolet, J. Boudon, A. Loiseau, S. Chevrier, R. Boidot, A. Oudot, B. Collin, E. Martin, P. A. Joy, N. Millot, G. Créhange, Docetaxel-titanate nanotubes enhance radiosensitivity in an androgen-independent prostate cancer model. Int J Nanomedicine 12, 6357-6364 (2017).
- Y. Bernhard, B. Collin, R. A. Decréau, Redshifted Cherenkov Radiation for in vivo Imaging: Coupling Cherenkov Radiation Energy Transfer to multiple Förster Resonance Energy Transfers. Sci Rep 7, 45063 (2017).
- N. Maindron, M. Ipuy, C. Bernhard, D. Lhenry, M. Moreau, S. Carme, A. Oudot, B. Collin, J. M. Vrigneaud, P. Provent, F. Brunotte, F. Denat, C. Goze, Near-Infrared-Emitting BODIPY-trisDOTA(111) In as a Monomolecular Multifunctional Imaging Probe: From Synthesis to In Vivo Investigations. Chemistry 22, 12670-12674 (2016).
- Laurent, C. Bernhard, S. Dufort, G. Jiménez Sánchez, R. Bazzi, F. Boschetti, M. Moreau, T. H. Vu, B. Collin, A. Oudot, N. Herath, H. Requardt, S. Laurent, L. Vander Elst, R. Muller, M. Dutreix, M. Meyer, F. Brunotte, P. Perriat, F. Lux, O. Tillement, G. Le Duc, F. Denat, S. Roux, Minor changes in the macrocyclic ligands but major consequences on the efficiency of gold nanoparticles designed for radiosensitization. Nanoscale 8, 12054-12065 (2016).
- D. Lhenry, M. Larrouy, C. Bernhard, V. Goncalves, O. Raguin, P. Provent, M. Moreau, B. Collin, A. Oudot, J. M. Vrigneaud, F. Brunotte, C. Goze, F. Denat, BODIPY: A Highly Versatile Platform for the Design of Bimodal Imaging Probes. Chemistry 21, 13091-13099 (2015).
- M. Jodoin, F. Pinheiro, A. Oudot, A. Lalande, Left-Ventricle Segmentation of SPECT Images of Rats. IEEE Trans Biomed Eng 62, 2260-2268 (2015).
- Duheron, M. Moreau, B. Collin, W. Sali, C. Bernhard, C. Goze, T. Gautier, J. P. Pais de Barros, V. Deckert, F. Brunotte, L. Lagrost, F. Denat, Dual labeling of lipopolysaccharides for SPECT-CT imaging and fluorescence microscopy. ACS Chem Biol 9, 656-662 (2014).
- De Taddéo, B. Collin, H. Hardy, B. Guichard, P. Trouilloud, O. Trost, [Can one harvest a long bone stick in the radial forearm flap? Original radioanatomical and NanoSPECT-CT Bioscan microvascular study]. Ann Chir Plast Esthet 59, 170-176 (2014).
- Y. Bernhard, B. Collin, R. A. Decréau, Inter/intramolecular Cherenkov radiation energy transfer (CRET) from a fluorophore with a built-in radionuclide. Chem Commun (Camb) 50, 6711-6713 (2014).
- J. M. Vrigneaud, A. Courteau, J. Ranouil, L. Morgand, O. Raguin, P. Walker, A. Oudot, B. Collin, F. Brunotte, Application of the optically stimulated luminescence (OSL) technique for mouse dosimetry in micro-CT imaging. Med Phys 40, 122102 (2013).
- F. Végran, R. Mary, A. Gibeaud, C. Mirjolet, B. Collin, A. Oudot, C. Charon-Barra, L. Arnould, S. Lizard-Nacol, R. Boidot, Survivin-3B potentiates immune escape in cancer but also inhibits the toxicity of cancer chemotherapy. Cancer Res 73, 5391-5401 (2013).
- Moreau, O. Raguin, J. M. Vrigneaud, B. Collin, C. Bernhard, X. Tizon, F. Boschetti, O. Duchamp, F. Brunotte, F. Denat, DOTAGA-trastuzumab. A new antibody conjugate targeting HER2/Neu antigen for diagnostic purposes. Bioconjug Chem 23, 1181-1188 (2012).